Hi, what do you want to do?
National Institute of Open Schooling
General Characteristics of the p-Block Elements
The 20th installment in a series of 36 focuses on the characteristics of the p-block elements. Learners discuss, read about, and answer questions pertaining to the occurrence of these elements in nature, their electron configurations,...
Rensselaer Polytechnic Institute
Molecules to the Max!—Teacher's Discovery Guide
Molecules to the Max! refers to a movie released in 2009 about the world of atoms and molecules. A helpful discovery guide provides five posters on science topics typically covered at the middle school level. It also explains the...
National Institute of Open Schooling
p-Block Elements and Their Compounds – II
Ozone, made of three bonded oxygen atoms, is found 15-30 km above Earth, has a strong smell, is blue, and blocks sunlight from hitting the surface of Earth. The 22nd lesson in a series of 36 specifically focuses on the important elements...
Chapman University
The Standard Model Poster
Chemistry classes will appreciate this color-coded, single-page reference sheet for The Standard Model of particle physics. It is divided into two main sections: elementary particles and compound particles, both with their antiparticles....
It's About Time
Chemical Names and Formulas
Abracadabra! Provide your class with the tools to perform a chemical "magic show" as they predict the charges of various ions, determine ionic compound formulas, and make observations to determine when a chemical reaction between...
Simon Fraser University
Chem1 Virtual Textbook: Organization of the Periodic Table
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site focuses specifically on the Periodic Table of Elements with information on the table's organization methods along with...
Other
Laws of Physics: Atoms and the Periodic Table
An overview of the differences between the metal and non metal groups. Includes a lot of information on the periodic table and has a plethora of graphs.
Simon Fraser University
Chem1 Virtual Textbook: The Limits of Classical Physics
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site discusses the limits associated with classical physics. Topics covered include light and heat with additional information...
Simon Fraser University
Chem1 Virtual Textbook: Sizes of Atoms and Ions
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site seeks to answer the question, What do we mean by the "size" of an atom? Terms such as metallic radius, covalent radius,...
Simon Fraser University
Chem1 Virtual Textbook: The Bohr Atom
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site discusses Niels Bohr and his work with the atom. Topics covered in the discussion include the atom before Bohr, Bohr's...
Simon Fraser University
Chem1 Virtual Textbook: The Shell Model of the Atom
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site discusses the properties of the atoms individually in relation to the main group elements of the Periodic Table.
Thomas Jefferson National Accelerator Facility
Jefferson Lab: It's Elemental Periodic Table of Elements
This page displays the Periodic Table of Elements and allows you to click each individual element for a page that displays in-depth information about that particular element. There are also links to many different games and learning...
Simon Fraser University
Chem1 Virtual Textbook: The Quantum Atom
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site specifically addresses the quantum atom and related topics. The related topics include the wave function and its physical...
Simon Fraser University
Chem1 Virtual Textbook: Periodic Trends in Ion Formation
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site discusses electron affinity and ionization energy in relation to ion formation. Charts and graphs are included as well.
Simon Fraser University
Chem1 Virtual Textbook: Spectrum of the Hydrogen Atom
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site discusses the hydrogen atom and its relation to spectrum. Included in the discussion is information on the Bohr model...
Simon Fraser University
Chem1 Virtual Textbook: The Uncertainty Principle
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site discusses the uncertainty principle and its association with Werner Heisenberg.
Simon Fraser University
Chem1 Virtual Textbook: Matter and Energy United
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site discusses the relation between matter and energy with attention given to Mass-energy, energy, and relativity.
Simon Fraser University
Chem1 Virtual Textbook: Spectrum of a Guitar String
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site discusses spectrum in relation to Bohr's model. Included in the topics covered are standing waves, boundary condition,...
Simon Fraser University
Chem1 Virtual Textbook: Electronegativity
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site discusses shared chemical bonds between two elements resulting in electronegative and electropositive outcomes.
Science4Fun
Science4 Fun: Elements
Fun and interesting information about elements. Learn about the periodic table, how they are distinguished, and the different families of elements.
Simon Fraser University
Chem1 Virtual Textbook: Wave, Particle, or What?
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site discusses light, waves, and particles. Part of the discussion involves a definition and working information on the...
Other
Chemguide: Electronegativity
This page explains what electronegativity is, and how and why it varies around the Periodic Table. It looks at the way that electronegativity differences affect bond type and explains what is meant by polar bonds and polar molecules.
Wikimedia
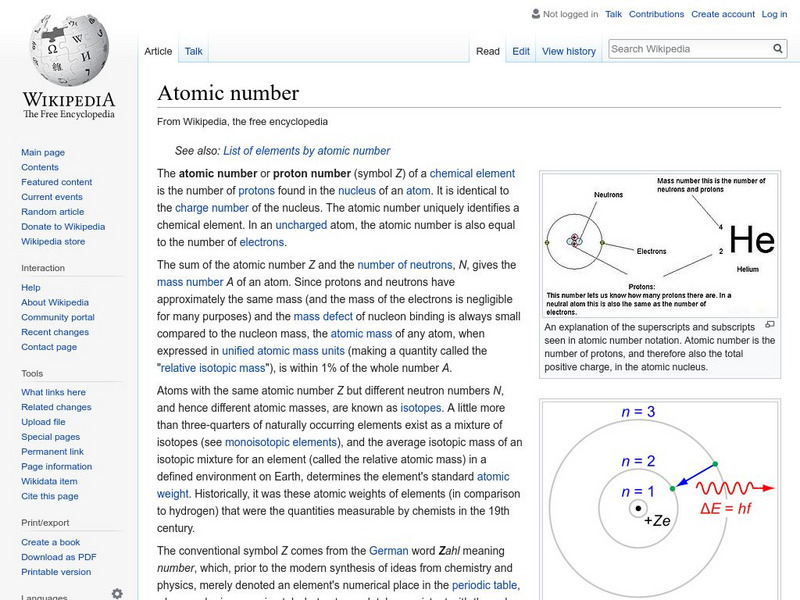
Wikipedia: Atomic Number
Wikipedia provides the definition of the term, "Atomic number," a term used in chemistry and physics to represent the number of protons in the nucleus of an atom.